Why are Blood Products Irradiated?
Blood products will sometimes need to be irradiated for certain populations of patients. Irradiation exposes the blood products to a specific amount of radiation for a specific portion of time, known as a dosage. The target dosage for blood product irradiation is 25Gy(Gray) or 2500 rads. The most common forms of blood irradiation are Xray irradiation and Gamma-ray irradiation (from radioactive cesium or cobalt). The US government is spearheading an initiative to replace Gamma-ray irradiators with Xray to remove the use of radioactive materials where possible.
Is irradiated blood radioactive?
Regardless of the method used, the process of irradiating blood products does NOT impart radioactivity to the product.
Irradiation Stickers
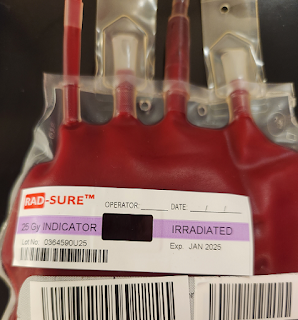 |
| A Red Cell unit that has been successfully irradiated. The "NOT" has disappeared, leaving only the words "IRRADIATED" on the righthand side. |
Graft-vs-Host Disease
Irradiation protects certain populations from experiencing what is known as TA-GVHD or Transfusion Associated Graft vs Host Disease. This happens when T-lymphocytes (a type of white blood cell) that remain in a donor bag after leukoreduction recognize the recipient's tissues as foreign and mount an immune response towards the recipient's tissues.
Almost all blood products in this day and age are leukoreduced. This means that the white blood cell content of a unit has been drastically reduced through a filtration process. The process of leukoreduction removes MOST white blood cells, but NOT all.
This cascade of an immune response associated with TA-GVHD has an exceedingly high mortality rate (>90%) so it is very important to recognize the populations who may need irradiated products.
In normal healthy "immunocompetent" individuals with a functioning immune system, the small amount of T-lymphocytes remaining would not be a problem. The recipient's immune system would recognize the donor's lymphocytes as foreign, and remove/destroy them before they have a chance to form a reaction. In patients who are immunocompromised or otherwise don't have a properly functioning immune system, it is possible that this destruction of donor lymphocytes would not happen.
Irradiation inactivates lymphocytes and prevents them from proliferating and mounting a response against the recipient's tissues.
If TA-GVHD occurs, it is almost certainly fatal. Greater than 90% of patients will die once the process begins, so it's VERY important to ensure irradiated blood products are given to those who need it.
Generally, only Red Blood Cells, Platelets, and Granulocytes are irradiated. Frozen products that are thawed before use like Plasma/FFP and Cryoprecipitate typically are not, because most cells do not survive the freezing process.
Irradiated Blood Product Indications
There are numerous indications for the need for irradiated blood products.
Any HLA-matched or HLA selected platelets
This is due to the fact that lymphocytes in very closely matched HLA products may evade the immune system of an immunocompetent recipient and still amount an immune response to tissues that are not a 100% perfect match.Directed Donations from 1st or 2nd degree relatives
Similar to the previous reason, if a patient receives a blood product from a close blood relative they may have very similar Human Leukocyte Antigen profiles. It is unlikely they will match 100%, but even 80-90% match could be enough to evade the immune system of the recipient. The donor lymphocytes can then react and start to attack based off of the 10-20% incompatibility causing TA-GVHD.Any granulocyte transfusion
Due to the fact that there are numerous, likely immunocompetent, T-lymphocytes in the unit that can mount a response. Irradiation generally does not affect the neutrophils in the unit, which is the main purpose of granulocyte transfusionBone Marrow Transplant or Candidate
Hematologic Malignancies
Such as Leukemia/Lymphoma/Multiple Myeloma/etcAplastic Anemia
Purine Analog and Antagonist Drugs
These type of drugs are used often in transplant and lymphocytic leukemia patients. Many ultimately act on the ability of the cells to perform DNA synthesis. This also causes a decrease in functional lymphocytes (which can then lead to TA-GVHD if transfused with non-irradiated products). Some of these drugs include Azathioprine, Bendamustine, Cladribine, Clofarabine, Deoxycoformicin, Fludarabine, Mercaptopurine, Pentostatin, and Thioguanine.Alemtuzumab
Also known as Campath or Lemtrada. It's a monoclonal antibody treatment that binds CD-52 on mature lymphocytes and targets them for destruction. Since this lowers and inhibits lymphocyte activity, this puts the patient at risk for TA-GVHD if given un-irradiated products.Anti-Thymocyte Globulin
Patients taking ATG, also known as Thymoglobulin or Atgam are at risk of TA-GVHD if not given irradiated products. This is because ATG is typically given to organ transplant patients to ward off organ rejection by inhibiting and removing healthy T-lymphocytes from circulation.Congenital Immunodeficiencies
Patients with conditions such as Severe Combined Immunodeficiency of DeGeorge syndrome should receive irradiated products as they have a genetic condition causing them to be immunodeficient and relatively unable to fight off immunocompetent donor lymphocytes.Premature/Low Birthweight (less than 1.2Kg) Neonates
These population of blood product recipients should receive irradiated blood as well, due to the fact that their immune systems are likely underdeveloped and many not function appropriately in response to donor lymphocytes.
Pathogen Inactivation
The system works by adding a psoralen-type chemical known as amotosalen to the platelet bag. The bag is then exposed to UV-A light. This causes the amotosalen to crosslink with nucleic acids. This interrupts the normal operation of viruses, bacteria, parasites, and T-lymphocytes that can cause TA-GVHD. Nearly all of the amotosalen is removed from the bag post-processing, and the unit can be sent to a Transfusion Center.
This treatment is an FDA approved substitute for physically irradiating a product, thus all amotosalen/UVA pathogen reduced platelets can, for all intents and purposes, be considered irradiated, because the causative agent of TA-GVHD is functionally inactivated. The units should NOT be physically irradiated at the Transfusion Center Blood Bank. Some Blood Centers have already switched to using this method on a majority of their platelets.

